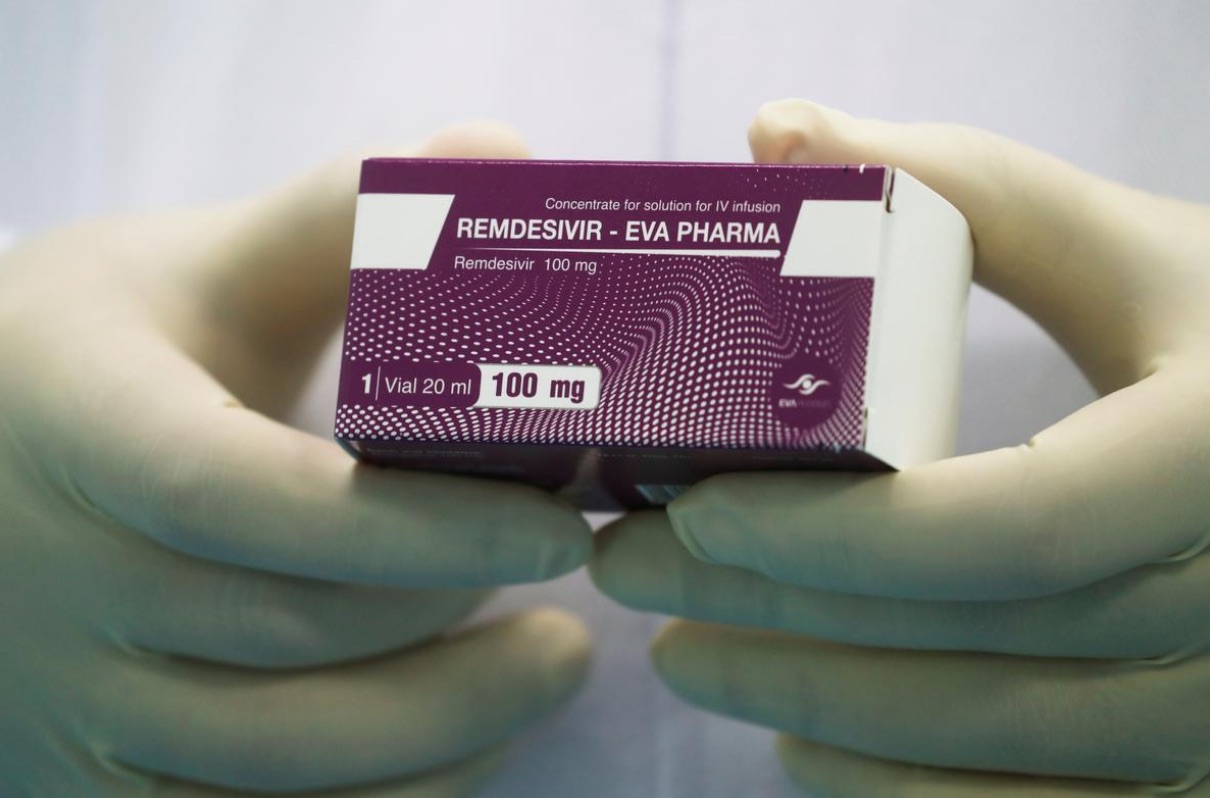
CAIRO, June 25 (Reuters) — Egypt’s Eva Pharma said on Thursday it had begun producing Remdesivir and Favipiravir, two drugs used in the treatment of patients with COVID-19.
Remdesivir is indicated for moderate to severe cases of COVID-19 for patients in intensive care who require oxygen, while Favipiravir is indicated for mild to moderate cases.
Eva Pharma, a generic drugmaker established in 1997, said earlier this month it had received a license from Gilead Sciences Inc to make Remdesivir in Egypt and distribute it in 127 countries.
It will initially make 500,000 doses a month to be sold locally for less than 2,000 Egyptian pounds (US$124.07) each, said general manager Amgad Talaat, adding that they had already started delivering the medicine to quarantine hospitals.
It has also started producing one million Favipiravir tablets a month, which will be on the market within two to three weeks, Talaat said. The price has still to be finalised.
Japan’s Fujifilm Holdings Corp, which makes Favipiravir under the brand name Avigan, said this month its research on the drug as a potential COVID-19 treatment may need to continue until July.
Eva Pharma said it had received permission from Egypt’s pharmaceutical regulatory authority to produce the drugs, and the body would decided whether they could be sold in pharmacies or only used in hospitals.
Talaat said that once Egyptian needs were met, Eva Pharma hoped to sell any excess doses to other Middle Eastern and African countries.
Egypt’s Rameda Pharmaceutical also said this month it had begun making Favipiravir under the brand name Anviziram and had approval to manufacture Remedsivir.
___
Reporting by Ehab Farouk; Writing by Patrick Werr; Editing by Kirsten Donovan
Image: A lab technician holds the coronavirus disease (COVID-19) treatment drug “Remdesivir” at Eva Pharma Facility in Cairo, Egypt June 25, 2020. (REUTERS/Amr Abdallah Dalsh)